In 2013, Professor Urban Alehagen and a team of researchers reported on the beneficial effects of daily supplementation of elderly Swedish citizens with a combination of 200 mg of Coenzyme Q10 and 200 mcg of selenium, in divided doses with meals, over a four-year period.

I am Jan Aaseth, a Norwegian medical doctor and university professor and researcher. In my career, I have specialized in medical biochemistry and in internal and occupational medicine. Much of my research focuses on the importance of micronutrient status throughout all stages of life. In recent years, I have investigated the role of selenium and coenzyme Q10 deficiency in the development of cardiovascular disease, with particular emphasis on oxidative stress and low-grade systemic inflammation.
Compared to the study participants in the placebo group, the KiSel-10 study participants who received the active treatment had significant heart health benefits [Alehagen 2013]:
- reduced risk of death from heart disease
- better heart function as shown on echocardiograms
- lower blood levels of the NT-proBNP protein, a bio-marker for the risk of heart failure
In 2015, Professor Alehagen and I analyzed what we knew, as clinicians, about selenium and Coenzyme Q10 and heart disease [Alehagen & Aaseth 2015]. We asked ourselves why that combination had protected against heart disease in elderly people.
Coenzyme Q10 and Selenium and Heart Disease
To start, we knew from the research literature that ischemic heart disease and heart failure are two conditions where increased oxidative stress plays a role. More about oxidative stress in a moment.
The results of the KiSel-10 study indicated that there exists an interrelationship between selenium and Coenzyme Q10, which makes it beneficial to administer them to populations with deficiencies of both.
Reactive Oxygen Species and Antioxidants
First, it is important to give some background information about oxidative stress and antioxidants.
Reactive oxygen species – popularly referred to as “free radicals” – are natural products of cell metabolism. The process of aerobic metabolism in the mitochondria seems to be a primary source of reactive oxygen species production [Alehagen & Aaseth 2015].
The cell signaling role of moderate numbers of reactive oxygen molecules contributes to good health and involves participation in the biosynthesis of complex organic molecules, detoxification of invasive chemical substances, and protection against pathogens such as bacteria and viruses [Nordman 2003].
Oxidative Stress = Imbalance of Free Radicals and Antioxidants
The term “oxidative stress” is used to describe a state in the cells in which reactive oxygen molecules are present in far greater numbers than are needed for acting as cell signaling molecules. When the reactive oxygen molecules exist in excessively high levels, they are strongly associated with the development of degenerative diseases and perhaps even with the aging process [Nordman 2003].
Typically, the thing that contributes to the volatility of the reactive oxygen molecules is that they are seeking to attract and accept one or two electrons from other molecules. Whenever they steal electrons from molecules that then become unstable and seek to attract other electrons, they can start a harmful chain reaction and cause oxidative damage to the cells and their DNA, proteins, and lipids.
Consequently, our cells need the means to neutralize these reactive oxygen species that are produced in excess of the need for the beneficial cell signaling functions. The molecules that can safely scavenge free radicals are called antioxidants.
Antioxidants are molecules in their reduced state, the state in which they have electrons that they can donate without becoming unstable themselves.
Coenzyme Q10 and Selenoproteins with Antioxidant Effects
The reduced state of Coenzyme Q10 molecules, called ubiquinol, and some of the selenium containing selenoproteins such as the glutathione peroxidases, the thioredoxin reductases, and selenoprotein P protect against oxidative stress.
- Ubiquinol protects against lipid peroxidation and also reduces the inflammatory response.
- The glutathione peroxidases, the most studied seleno-enzymes, donate electrons to hydrogen peroxide molecules and catalyze H2O2’s reduction to water and oxygen; they also catalyze the reduction of other peroxide radicals to alcohols and oxygen.
- The seleno-enzymes, the thioredoxin reductases, are the most efficient enzymes for reducing molecules in the the ubiquinone state of Coenzyme Q10, thus regenerating the lipid-soluble antioxidant ubiquinol.
Regenerating Ubiquinol with the Help of Selenium Containing Seleno-enzymes
Once the ubiquinol molecules donate electrons to quench reactive oxygen molecules, they themselves are oxidized. They have no electrons to donate and are stable. They become electron acceptors.
To replenish the supply of the antioxidant ubiquinol in the cells, it is necessary for some of these molecules in their oxidized state – called ubiquinone – to be regenerated in their reduced state – the electron donor state.
This is important because the mitochondria – those bean-shaped energy factories in the cells – are very vulnerable to oxidative damage. In heart disease, reactive oxygen species in the mitochondria and in the cytosol of the cells become distorted, leading to an oxidative imbalance, to mitochondrial dysfunction, and eventually to cell death [Alehagen & Aaseth 2015].
Reduced Reserve of Ubiquinol in Elderly People
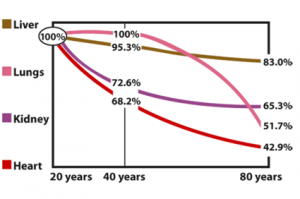
A study done by Kalén, Appelkvist, and Dallner has shown that Coenzyme Q10 content peaks in many organs at 20 years of age and is characterized by a continuous decline during further aging.
Elderly people may have a reduced reserve of ubiquinol for two reasons:
1. The cells’ endogenous synthesis of Coenzyme Q10 declines with increasing age such that an 80-year-old person may synthesize only one half the amount of the Coenzyme Q10 that a 20-year-old person does [Kalen 1989]. Elderly people synthesize less ubiquinone; consequently, there is less ubiquinone to be used for energy production and to be converted to the antioxidant state, ubiquinol.
2. Elderly people living in a region with low selenium content in the soil and the food may have less selenium and fewer selenoproteins available to regenerate ubiquinol from the available ubiquinone.
The level of oxidative stress is elevated in heart disease patients. In addition, high levels of systemic inflammation are associated with high levels of oxidative stress [Alehagen & Aaseth 2015].
Thioredoxin Reductase Regenerating Ubiquinol from Ubiquinone
Ubiquinone – the oxidized state of Coenzyme Q10 – is essential to the production of ATP energy in the mitochondria. In the process of bio-energetics, the ubiquinone is reduced to ubiquinol.
When the ubiquinol molecules donate electrons to protect against oxidative damage, they revert back to their oxidized state – ubiquinone. The question then becomes, how to regenerate the antioxidant state of Coenzyme Q10.
One of the important interrelationships between selenium and Coenzyme Q10 (ubiquinone) is the catalytic role of selenoproteins such as the thioredoxin reductases in the metabolic conversion of ubiquinone to ubiquinol.
Xia et al. [2003] showed that thioredoxin reductase is the most efficient enzyme that reduces ubiquinone to ubiquinol.
Coenzyme Q10 Needed for the Synthesis of the Antioxidant Seleno-Enzymes
Just as we need sufficient quantities of selenoproteins to regenerate ubiquinol, so too do we need adequate quantities of Coenzyme Q10 for the optimal synthesis of the selenocysteine-containing enzymes [Moosmann & Behl 2004].
In our 2015 article, Prof. Alehagen and I explained that there exists a special interrelationship between Coenzyme Q10 and selenium containing selenoproteins as follows:
- A deficiency of selenium could restrict the cells’ ability to obtain optimal concentrations of Coenzyme Q10 in its antioxidant form.
- The cells are dependent on sufficient levels of Coenzyme Q10 for the optimal functioning of selenium
- Coenzyme Q10 plays an important part in the synthesis of selenocysteine, which is required for the synthesis of the antioxidant selenoproteins glutathione peroxidase, thioredoxin reductase, and selenoprotein P.
Importance of the Mevalonate Biological Pathway for Coenzyme Q10 and for Selenoproteins
Moosmann and Behl [2004] have shown that the synthesis of the selenocysteine containing selenoproteins proteins requires a functional mevalonate pathway.
Note: The mevalonate pathway is the biological pathway in which cholesterol, Coenzyme Q10, and dolichol are produced.
A side road of the mevalonate pathway is the enzymatic isopentenylation of selenocysteine-tRNA, the tRNA to decode the selenocysteine amino acid. The isopentenylation of the selenocysteine-tRNA requires isopentenyl pyrophosphate, which it gets from the mevalonate pathway.
If the mevalonate pathway is not functioning optimally, there will not be enough Coenzyme Q10 synthesized and not enough selenocysteine produced for incorporation into selenoproteins.
Selenium Needed for the Synthesis of Coenzyme Q10
We cannot deprive humans of selenium to see what effect selenium deficiency has on Coenzyme Q10 bio-synthesis.
Vadhanavikit and Ganther [1994] have done animal studies that show that selenium deficiency – animals fed a diet lacking selenium for 18 months – is associated with levels of Coenzyme Q9 and Coenzyme Q10 in the liver that were 40% and 67% of the levels in selenium-adequate animals, respectively. These results are similar to the findings using a shorter feeding period (4.5 months).
Vadhanavikit and Ganther speculated that a decreased ability of the cells to synthesize Coenzyme Q10 because of selenium deficiency is associated with a lack of glutathione peroxidase protection against oxidative stress in the cells.
Conclusion
- The seleno-enzyme thioredoxin reductase has been shown to be the most efficient enzyme for reducing ubiquinone and regenerating the lipid-soluble antioxidant ubiquinol.
- The selenoproteins – the glutathione peroxidases and the thioredoxin reductases – are themselves important antioxidants.
- Elderly people are likely to have reduced Coenzyme Q10 biosynthesis and reduced selenoprotein formation.
- Increased levels of oxidative stress – inadequate availability of antioxidants – are associated with increased risk of ischemic heart disease and heart failure.
Read our key article about CoQ10 and cardiovascular health in elderly people
References
Alehagen U, Johansson P, Björnstedt M, Rosén A, Dahlström U. Cardiovascular mortality and N-terminal-proBNP reduced after combined selenium and coenzyme Q10 supplementation: a 5-year prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. Int J Cardiol. 2013 Sep 1;167(5):1860-6.
Alehagen U, Aaseth J. Selenium and coenzyme Q10 interrelationship in cardiovascular diseases–A clinician’s point of view. J Trace Elem Med Biol. 2015;31:157-62.
Kalén A, Appelkvist EL, Dallner G. Age-related changes in the lipid compositions of rat and human tissues. Lipids. 1989 Jul;24(7):579-84.
Moosmann B, Behl C. Selenoproteins, cholesterol-lowering drugs, and the consequences: revisiting of the mevalonate pathway. Trends Cardiovasc Med. 2004;14:273–81.
Nordman T, Xia L, Björkhem-Bergman L, Björnstedt M, Olsson J. Regeneration of the antioxidant ubiquinol by lipoamide dehydrogenase, thioredoxin reductase and glutathione reductase. BioFactors. 2003;18(1-4):45-50.
Vadhanavikit S, Ganther HE. Selenium deficiency and decreased coenzyme Q levels. Mol Aspects Med. 1994;15 Suppl:s103-7.
Xia L, Nordman T, Olsson JM, Damdimopoulos A, Björkhem-Bergman L, Nalvarte I, Eriksson LC, Arnér ES, Spyrou G, Björnstedt M. The mammalian cytosolic selenoenzyme thioredoxin reductase reduces ubiquinone. A novel mechanism for defense against oxidative stress. J Biol Chem. 2003 Jan 24;278(4):2141-6.
The information presented in this review article is not intended as medical advice and should not be construed as such.
30 March 2021





Leave A Comment