
Coenzyme Q10 is a fat-soluble redox molecule that we produce in nearly all cells in the body. Once we reach our 20’s, our body’s synthesis of Coenzyme Q10 begins to decline with increasing age. But our need for Coenzyme Q10 continues, both in the process of producing ATP energy molecules and in the cells’ defense against harmful free radicals. Consequently, we need a good daily supplement.
NQO1 gene? … NQO-1 enzymes? … I don’t remember reading or hearing about the NQO-1 gene in all of the years that I have been taking Coenzyme Q10 supplements. But, for Dr. William Judy, the NQO-1 gene seems to be one of the three or four Q10 questions that he has been most preoccupied with for several years.
NQO-1 gene important in the biochemistry of Coenzyme Q10
NQO-1 is used as shorthand for both the gene and the oxidoreductase enzymes that the gene codes for. The NADPH-Quinone Oxidoreductase-1 gene – to give it its full name – is the gene that codes in humans for the production of enzymes that reduce quinones to hydroquinones.
Now, that sentence is a mouthful. What does it mean, and how does it relate to the controversy about the ubiquinone and ubiquinol forms of Coenzyme Q10? Let me take it the name segment by segment.
CoQ10 and the production of ATP energy molecules
Okay, it gets a bit complicated. First, we need to know that NAD+ (Nicotinamide Adenine Dinucleotide – oxidized form) and NADH (reduced form) are molecules that are important in the production of the ATP energy molecules in human cells.
Secondly, we need to know that NADP+ (Nicotinamide Adenine Dinucleotide Phosphate) is more or less an identical twin of NAD+ but with a phosphate group instead of an alcohol group attached. NADPH, then, is the reduced form of NADP+ (it has taken on electrons). NADPH is needed as a reducing agent (a reducing agent donates electrons) in lipid and nucleic acid synthesis.
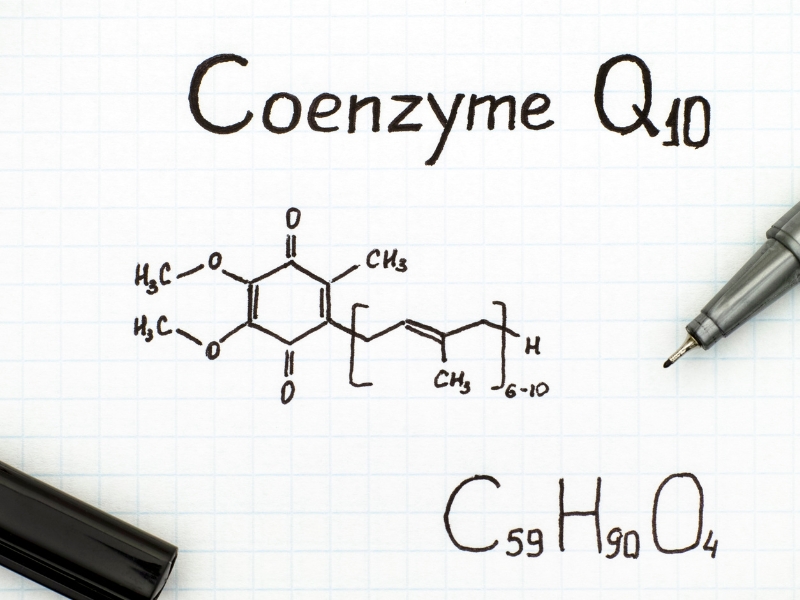
Coenzyme Q10 is a benzoquinone
Quinones are cyclic organic compounds that contain two carbonyl groups (carbonyl groups have a carbon atom double-bonded to an oxygen atom). Quinones are the oxidized form of redox molecules whose most characteristic chemical reaction is to be reduced (to take on 1 or 2 electrons). Typically, a benzoquinone is reduced to become a hydroquinone. The reduction is reversible. The hydroquinone can be and will be converted back to the benzoquinone by giving up the two electrons that it took on.

Dr. William Judy, SIBR Research Institute, has taught me much of the information that is presented in this article. Dr. Judy has done lab studies, animal studies, and clinical trials of the absorption, efficacy, and safety of Coenzyme Q10 for over 35 years.
Conversion from ubiquinone to ubiquinol with help from NQO-1 enzymes
In the specific case of Coenzyme Q10, the NQO-1 oxidoreductase enzymes facilitate the conversion of ubiquinone (the oxidized form of Q10) to ubiquinol (the reduced form). Because Coenzyme Q10 molecules are redox molecules, the ubiquinol can and will be converted back to ubiquinone.
Ubiquinol’s antioxidant function
However, the conversion of ubiquinol back again to ubiquinone is not thought to take place through the catalysis of the NQO-1 oxidoreductase enzymes. Ubiquinol holds its two extra electrons rather loosely, and it gives these two electrons up readily to harmful free radicals. Giving up its two electrons to neutralize free radicals is what makes the ubiquinol into the oxidized form again, the ubiquinone form.
So, big picture:
- NQO-1 oxidoreductase enzymes help convert ubiquinone to ubiquinol
- Neutralizing harmful free radicals converts ubiquinol back to ubiquinone
Coenzyme Q10 and oxidative stress and free radicals
A word about free radicals. Free radicals are a product of the use of oxygen in the body. Often, they are referred to by the term “reactive oxygen species,” abbreviated ROS. They are reactive molecules containing oxygen atoms: peroxide radicals, superoxide radicals, hydroxyl radicals, and singlet oxygen radicals. To become stable molecules, they need to take on electrons.
If the harmful free radicals cannot find donor molecules (like ubiquinol) from which to take extra molecules, then they cause damage to cell structures and to cell DNA. The damage caused by free radicals is called oxidative stress. The importance of ubiquinol is that it is admirably able to donate its extra electrons to the needy free radicals. In so doing, the ubiquinol is oxidized back to the ubiquinone form, which is the form that is important in the energy production process in the cells.
Coenzyme Q10 and free radicals and cancer
One theory of cancer (there is another theory, one favored by Dr. Karl Folkers … I will write more about it at a later time) is that when sufficient oxidative damage has occurred in cells, the DNA of the cells is damaged. When the cells’ DNA is damaged, then the cells lose control of their growth regulation mechanisms, and the cells become tumor cells instead of undergoing apoptosis (the name for programmed cell death). Thus, the antioxidant role of Coenzyme Q10 may be important in the chemoprevention of certain cancers.
Oxidoreductases are enzymes that catalyze (facilitate) the transfer of electrons from one molecule to another molecule. Mostly, reductase enzymes facilitate reductions (the accepting of electrons). One important reductase in the context of Coenzyme Q10 is the peroxidase enzyme, which catalyzes the reduction of the free radical hydrogen peroxide.
Coenzyme Q10 as ubiquinone and ubiquinol
Remember: the ubiquinone form of Q10 is the oxidized form. It is poised to take on two electrons in a redox reaction and become the ubiquinol form. The NADPH dehydrogenase quinone 1 enzyme that is encoded by the NQO-1 gene facilitates the conversion of the ubiquinone form of Q10 to the ubiquinol form of Q10.
An intermediate form of Coenzyme Q10
I have jumped from ubiquinone (the oxidized form, the form that has given up two electrons) to ubiquinol (the reduced form, the form that has taken on two electrons). There is an intermediate form, the ubisemiquinone form, the form that has taken on one electron. Ubisemiquinone is highly unstable, so it may not form at all, and, if it does take form, it does not last long. It quickly takes on another electron and becomes ubiquinol. Therefore, our focus is on ubiquinone and ubiquinol. Ubisemiquinone is not suitable for use as a supplement.
Redox conversions of Coenzyme Q10 molecules
In this article, my interest in NQO-1 is an interest in when and how conversions of the Coenzyme Q10 molecule take place. Later, in a separate article, I will want to look at when and how the expression of the NQO-1 gene differs in diverse populations.
From ubiquinone to ubiquinol to ubiquinone
There is some evidence now, from large animal (dog) studies, that Coenzyme Q10, if taken in the form of a ubiquinol supplement, will be converted to its ubiquinone form before it reaches the absorption cells in the small intestine, and it will be absorbed as ubiquinone.
Then, while the Coenzyme Q10 passes through the lymph and the blood, reaching a peak concentration in the blood in approximately 6 – 8 hours, it will be almost entirely in the ubiquinol form. One possible explanation for the presence in the stomach and small intestine as ubiquinone and the presence in the lymph and blood as ubiquinol is that the necessary NQO-1 oxidoreductase enzymes are not present in the tissues of the stomach and the small intestine but are present in the endothelial cells (squamous cells) that line the inner walls of the lymph and blood vessels.
Coenzyme Q10 from the blood to the cells
Of special interest is the manner in which the ubiquinol molecules leave the blood and enter the tissues and the cells. I do not know the answer to this question. But, it is clear that, at some time and place and manner, the ubiquinol must be converted to ubiquinone and must make it way to the mitochondria in the cells if it is to benefit the energy production process. Quite possibly, oxidation of the ubiquinol molecules by free radicals accounts for some of this conversion, which is, of course, beneficial to the cells in that the oxidation of ubiquinol protects against oxidative stress.
Bottom line
All the evidence we have at present points to starting with a well-formulated Coenzyme Q10 supplement in the form of ubiquinone and letting the enzymes and free radicals take care of the rest. For good heart health, we need an adequate daily intake of Coenzyme Q10. That older adults can take and benefit from Coenzyme Q10 supplements in the form of ubiquinone is well illustrated by the 443-person KiSel-10 clinical trial.
Sources:
Alehagen, U., Johansson, P., Björnstedt, M., Rosén, A., & Dahlström, U. (2013). Cardiovascular mortality and N-terminal-proBNP reduced after combined selenium and coenzyme Q10 supplementation: a 5-year prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. International Journal Of Cardiology, 167(5), 1860-1866.
Judy, W.V., Stogsdill, W.W., Judy, D.S., & Judy, J.S. (2007). Coenzyme Q10: Facts or Fabrications? Natural Products Insider. Retrieved from http://www.zmc-usa.com/docs/CoQ10_Facts_or_Fabrications.pdf.
Judy, W.V. (2016). Private communications.
Mortensen, S. A., Rosenfeldt, F., Kumar, A., Dolliner, P., Filipiak, K. J., Pella, D., & … Littarru, G. P. (2014). The effect of coenzyme Q10 on morbidity and mortality in chronic heart failure: results from Q-SYMBIO: a randomized double-blind trial. JACC. Heart Failure, 2(6), 641-649.
Mortensen, S. A. (2015). Coenzyme Q10: Will This Natural Substance Become a Guideline-Directed Adjunctive Therapy in Heart Failure? JACC. Heart Failure, 3(3), 270-271. doi:10.1016/j.jchf.2014.12.006






Some of us who had a personal genetic test done (23+Me) discover we have the NQO1 gene polymorphism C609T, said to reduce our ability to make ubiquinol from ubiquinone, the size of effect depending on whether homozygous or heterozygous. I have the heterozygous form which reduces conversion to 1/3 of normal. Actually I’ve been taking high-quality ubiquinone for years since discovering it stopped my gums bleeding, and had other health benefits (so I’m inclined to believe the report that I have a CQ10 problem). “Nutrigenomics” practitioners now tend to advise those of us with this C609T variant to take ubiquinol instead of ubiquinone. But having carefully read your website, it seems like this might be pointless because it’d be converted back to ubiquinone in the stomach. Can I ask what Dr Judy says about this? And if he has any advice for those of us with this allele?
Gilly,
I will ask Dr. Judy and will get back to you ASAP.
Richard